Why Does the Quality of NK Cells (Natural Immunity) Differ Between Providers? What Are the Causes?

Why Does the Quality of NK Cells (Natural Immunity) Differ Between Providers? What Are the Causes?
NK cells (Natural Killer cells) are a critical component of the innate immune system. Their primary function is to identify and eliminate virus-infected cells and cancerous cells. NK cells circulate in the bloodstream, continuously surveilling abnormal cells. Upon recognition, they release cytotoxic proteins, such as interferon-gamma (IFN-γ), to destroy abnormal targets and secrete signaling molecules that recruit other immune cells to help eliminate the threat.[1]
In normal individuals, NK cells account for approximately 5–10% of total circulating white blood cells. Although they represent a relatively small proportion, they play a vital role in cancer surveillance and antiviral defense. Studies have shown that individuals with higher NK cell functional activity tend to have lower cancer incidence rates.[2,3] Importantly, these findings emphasize NK cell function rather than simply NK cell count, explaining why measuring white blood cell numbers alone does not accurately reflect immune performance.
Because of their natural tumor-targeting ability, NK cells have been incorporated into cancer treatment strategies. This approach involves expanding and activating NK cells ex vivo before reinfusing them into the patient. Clinical studies have reported outcomes in various cancers, including colorectal cancer, gastric cancer, and prostate cancer.[4,5] One major factor influencing therapeutic outcomes is the functional readiness of NK cells at the time of reinfusion. The quality and activation status of NK cells depend heavily on the laboratory expansion method used.[1]
NK cell expansion can be performed using either feeder-cell-based methods or feeder-free methods. Feeder-cell-based culture techniques are popular because they can generate large numbers of NK cells within a relatively short time. However, one disadvantage is that NK cell function may decrease after reinfusion, as supportive feeder cells are no longer present. In contrast, feeder-free NK cell culture methods typically produce lower overall cell yields, but they may help maintain more stable functional activity after transplantation.
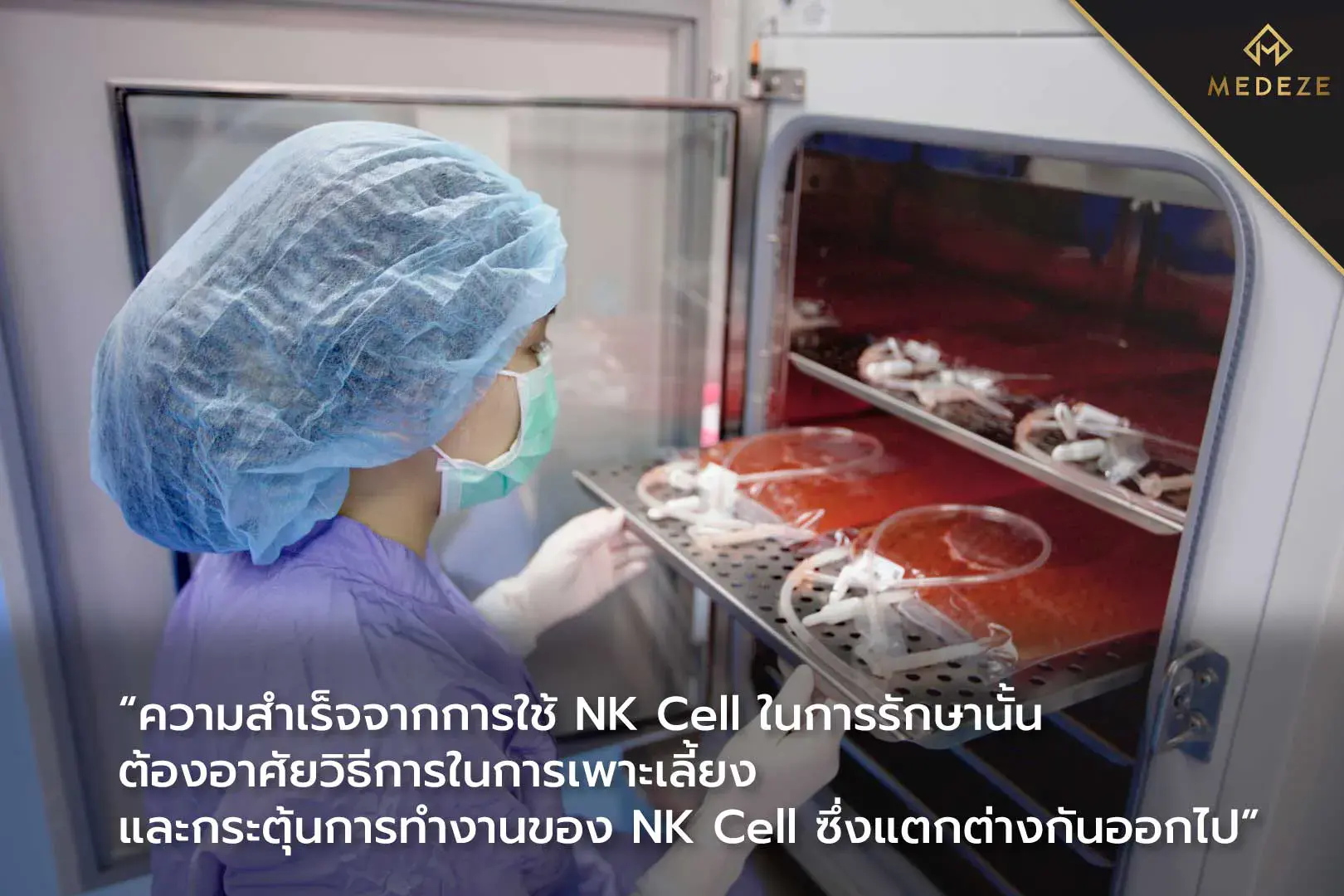
At MEDEZE, we implement a feeder-free NK cell expansion method known as the Osaki Method, developed by Junichi Masuyama, an immunologist at the New City Osaki Clinic. This method utilizes a proprietary culture medium within a patented closed-system platform. The formulation contains specific activating components that stimulate NK cell proliferation and enhance functional activation, resulting in NK cells that are immediately ready to perform upon reinfusion. Compared with conventional culture techniques, the Osaki Method improves both cell quantity and activation status.[6] NK cells expanded using the Osaki Method have been reported to increase activated NK cell numbers by approximately 510–1,500-fold.[6,7]

In Japan, NK cells expanded using the Osaki Method have been incorporated into standard treatments for patients with pancreatic, colorectal, lung, and gastric cancer.[8] There has also been a reported case in Thailand involving a late-stage lung cancer patient who did not respond to chemotherapy. Following treatment with NK cells activated through the Osaki Method, the patient’s clinical condition improved progressively.[5]
Through collaboration between Junichi Masuyama, developer of the Osaki Method at New City Osaki Clinic, and Dr. Veerapol Khemarangsan, founder of MEDEZE GROUP, the full NK cell expansion technology has been directly transferred to MEDEZE. This includes proprietary culture media, environmental control systems, and standardized protocols that enable the large-scale production of highly activated NK cells suitable for reinfusion, which is commonly referred to as Highly Active NK Cell Therapy.
References
1) Childs, R. W., & Berg, M. (2013). Bringing natural killer cells to the clinic: ex vivo manipulation. Hematology 2013, the American Society of Hematology Education Program Book, 2013(1), 234-246.
2) IMAI, Kazue, et al. Natural cytotoxic activity of peripheral-blood lymphocytes and cancer incidence: an 11-year follow-up study of a general population. The lancet, 2000, 356.9244: 1795-1799.
3) LEE, Saet-byul, et al. A high-throughput assay of NK cell activity in whole blood and its clinical application. Biochemical and biophysical research communications, 2014, 445.3: 584-590.
4) WU, Song-Yang, et al. Natural killer cells in cancer biology and therapy. Molecular Cancer, 2020, 19.1: 1-26.
5) MASUYAMA, Junichi, et al. NK cell therapy for end‑stage cancerous patient: A case study. South Asian journal of cancer, 2014, 3.02: 143-143.
6) MASUYAMA, Jun-ichi, et al. Ex vivo expansion of natural killer cells from human peripheral blood mononuclear cells co-stimulated with anti-CD3 and anti-CD52 monoclonal antibodies. Cytotherapy, 2016, 18.1: 80-90.
7) REZAEIFARD, Somayeh, et al. Autologous Natural Killer Cell-enrichment for Immune Cell Therapy: Preclinical Setting Phase, Shiraz Experience. Iranian Journal of Allergy, Asthma and Immunology, 2021, 1-11.
8) New City Osaki Clinic. Types of cancer and number of cases treated at the New City Osaki Clinic. [Online]. 2021. Available at: https://www.nco-clinic.jp/e/case/index.html. [August 2021].
Search Articles
Latest Articles
สายสะดือทารกแรกเกิด แหล่งรวมสเต็มเซลล์ ในการรักษาโรคและฟื้นฟูสุขภาพ
What are Mesenchymal Stem Cells (MSCs)? Exploring Their Benefits and Potential
What is Adipose Tissue? Can Fat Tissue Really Help with Anti-Aging?
Understanding Modern Innovation: NK Cells in Eliminating Cancer Cells and Virus-Infected Cells
What are NK Cells? The Immune Cells that Combat Viruses and Cancer Cell
