Stem Cell Storage at Any Age with Global Standards by MEDEZE

Stem Cell Storage at Any Age with Global Standards by MEDEZE
The medical use of stem cells, also known as progenitor cells, continues to expand rapidly. Beyond applications in blood disorders, certain cancers, and inherited metabolic diseases, stem cell therapy is now being explored for congenital diabetes, brain injury recovery, immune-related diseases, and even cardiovascular conditions.[1] Stem cells used in cell-based therapies can be derived from several sources. The most widely accepted sources today include umbilical cord blood, umbilical cord tissue, peripheral blood and bone marrow, and adipose (fat) tissue. Among these, umbilical cord blood and umbilical cord tissue stem cells offer significant advantages, as collection does not interfere with the newborn or the mother. The cells are obtained from the placenta and umbilical cord after delivery and are considered the most youthful and biologically potent, retaining a high capacity for cell division. Multiple studies have demonstrated that stem cell proliferation decreases with age at the time of collection, highlighting the importance of early, properly managed stem cell storage.[2]

Stem Cell Banks in Thailand
The laboratories that can store stem cells include both government-operated institutions, such as the Thai Red Cross Society,[3] and private stem cell laboratories. Public institutions may face limitations in accessibility, eligibility criteria, and donor-recipient compatibility. Private stem cell banks offer greater flexibility and convenience, as individuals can store and later use their own stem cells. This significantly reduces the risk of immune rejection and minimizes the possibility of cross-contamination or infectious transmission.
Two Key Factors That Determine Successful Stem Cell Storage
For private laboratories offering stem cell banking services, compliance with international laboratory standards is essential. Facilities must be fully certified for the processing, cultivation, cryopreservation, and long-term storage of all stem cell types, including blood- and tissue-derived stem cells. The primary objective for clients is to ensure that stem cells remain viable over extended periods and can be successfully revived and used when needed. Therefore, individuals considering stem cell storage should carefully evaluate laboratories against two critical standards.

Cell Isolation Process
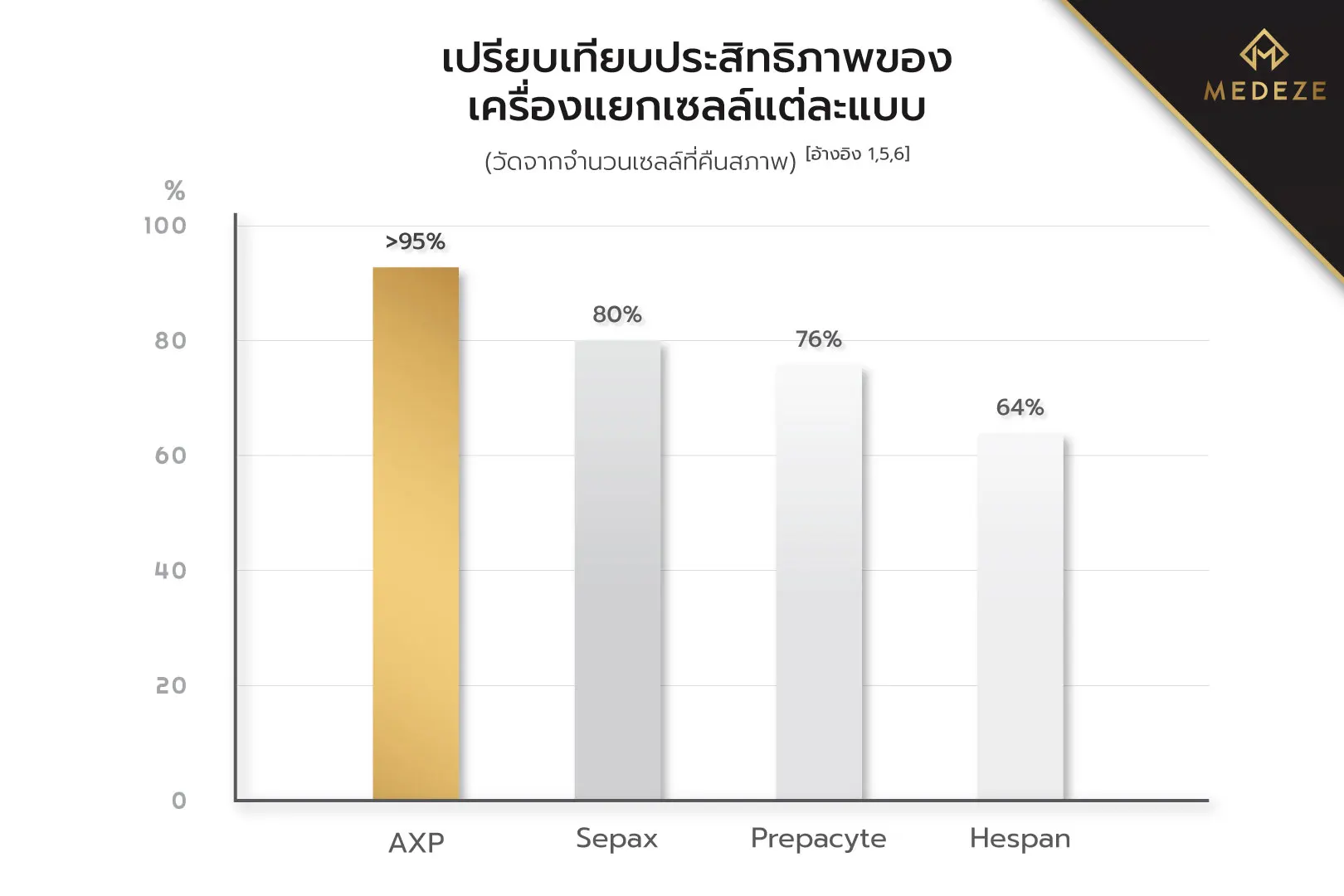
Blood Stem Cell IsolationFor Blood Stem Cell Isolation, several methods are available. However, the most advanced and globally recognized technique is isolation using the AXP® (AutoXpress Platform). AXP® is a fully automated blood stem cell separation system that significantly reduces human error and operates within a closed system, allowing precise environmental control while minimizing the risk of external contamination. In Thailand, MEDEZE is the only laboratory that utilizes the AXP® system for stem cell isolation. This is the same technology used by leading global institutions, including The New York Blood Center, the largest blood bank in the United States.[4] When compared with other commonly used isolation methods, such as Hespan, Prepacyte, or Sepax, which all demonstrate lower post-thaw cell recovery rates, AXP® consistently delivers superior outcomes.[1,5,6] The cell recovery rate achieved with AXP® exceeds 95%, significantly higher than alternative methods, which typically yield recovery rates of no more than 80%.
Tissue-Derived Stem Cell Isolation For tissue-derived stem cells, MEDEZE performs immediate cell isolation upon receipt of the tissue, carried out by highly trained specialists in stem cell processing. The isolated cells are then expanded using the Quantum automated cell culture system, which enables rapid cell proliferation while preserving the stem cells' youthfulness and regenerative potential at the chromosomal level.
Stem Cell Storage Process
Blood Stem Cell Isolation Blood Stem Cell Cryopreservation is a critical step in stem cell banking, as clients expect their stem cells to remain viable, contamination-free, and clinically usable over the long term. MEDEZE operates laboratories that meet Cleanroom Class 100 standards, enabling clinical-grade tissue processing under highly controlled, sterile conditions. For long-term storage, MEDEZE uses a nitrogen-vapor-phase cryogenic freezing system at –196°C, which maintains stem cell integrity throughout the 60-year storage period in a fire-resistant vault.
Based on the standards mentioned, MEDEZE has earned international accreditation from the American Association of Blood Banks (AABB) for cord blood stem cell processing, long-term storage, and global transportation for clinical use, in accordance with the National Environmental Balancing Bureau (NEBB) laboratory quality standards from Germany.
References
1) Willert J, Purdon T, Harris D. Umbilical cord blood—Biology, banking, and therapeutic use. US Obstetrics and Gynecology. 2008;3(1):68-72.
2) Harris DT. Stem cell banking for regenerative and personalized medicine. Biomedicines. 2014 Mar;2(1):50-79.
3) ศูนย์บริการโลหิตแห่งชาติ สภากาชาดไทย. การบริจาคสเต็มเซลล์. [Online]. Available at: https://blooddonationthai.com/?page_id=784. [August 2021]
4) Cord Blood Registry. Cord Blood Registry Achieves Industry-Leading Stem Cell Recovery With New Automation Technology. [Online]. 2007. Available at: https://www.biospace.com/article/releases/-b-cord-blood-registry-b-achieves-industry-leading-stem-cell-recovery-with-new-automation-technology-/. [July 2021]
5) Rubinstein P. Cord blood banking for clinical transplantation. Bone marrow transplantation. 2009 Nov;44(10):635-42.
6) Basford C, Forraz N, Habibollah S, Hanger K, McGuckin CP. Umbilical cord blood processing using Prepacyte‐CB increases haematopoietic progenitor cell availability over conventional Hetastarch separation. Cell proliferation. 2009 Dec;42(6):751-61.
Search Articles
Latest Articles
สายสะดือทารกแรกเกิด แหล่งรวมสเต็มเซลล์ ในการรักษาโรคและฟื้นฟูสุขภาพ
What are Mesenchymal Stem Cells (MSCs)? Exploring Their Benefits and Potential
What is Adipose Tissue? Can Fat Tissue Really Help with Anti-Aging?
Understanding Modern Innovation: NK Cells in Eliminating Cancer Cells and Virus-Infected Cells
What are NK Cells? The Immune Cells that Combat Viruses and Cancer Cell
