Where Can Stem Cell Storage Be Done in Thailand? Important Considerations and Precautions
Where Can Stem Cell Storage Be Done in Thailand? Important Considerations and Precautions
Nowadays, the term "stem cell" has become widely recognized. Numerous research studies have demonstrated the benefits and value of stem cells, making stem cell storage increasingly popular both in Thailand and internationally.
Stem cells are unique in that they do not have a specific function initially. They can divide to produce identical cells and have the potential to differentiate into various specialized cell types. They play a crucial role in repairing and regenerating the body. Therefore, stem cells are suitable for promoting longevity, anti-aging, and are valuable assets in fighting various diseases. There are two main types of stem cells: Hematopoietic Stem Cells (HSCs) and Mesenchymal Stem Cells (MSCs).
Hematopoietic Stem Cells (HSCs) are blood-forming stem cells. They not only have the property of dividing into blood system cells but can also differentiate into endothelial cells (blood vessel cells), making them useful for various applications.

Mesenchymal Stem Cells (MSCs) are tissue stem cells that can differentiate into cartilage cells, lung cells, liver cells, pancreatic cells, kidney cells, and other cells in the body. A special property is their ability to home in on areas of inflammation and produce substances that help repair tissues, reduce inflammation, and balance the immune system.
The advantages of Mesenchymal Stem Cells (MSCs) are their robustness and ability to be cultured to increase their numbers. There is extensive research on their use in humans to treat conditions such as osteoarthritis, diabetes, liver cirrhosis, SLE (Lupus), stroke, and many others.
Where to Store Stem Cells in Thailand
Currently, stem cell storage services in Thailand are available through both public and private institutions.

Each stem cell storage facility has its own pros and cons, such as facilities, specific medical techniques, storage technologies, stem cell experts, collection sites, and pricing. Therefore, choosing a storage facility requires research to find the best fit for yourself. However, the most critical factors to consider are safety and cleanliness. Medeze Group is a leading innovator in the stem cell industry, with a team dedicated to creating advanced technologies to care for your stem cells with the highest standards of cleanliness and safety.

Stem cell collection and processing must be performed by experts in specialized laboratories, as safety is the primary concern. Humans can collect Hematopoietic Stem Cells (HSCs) from umbilical cord blood, bone marrow, or peripheral blood.
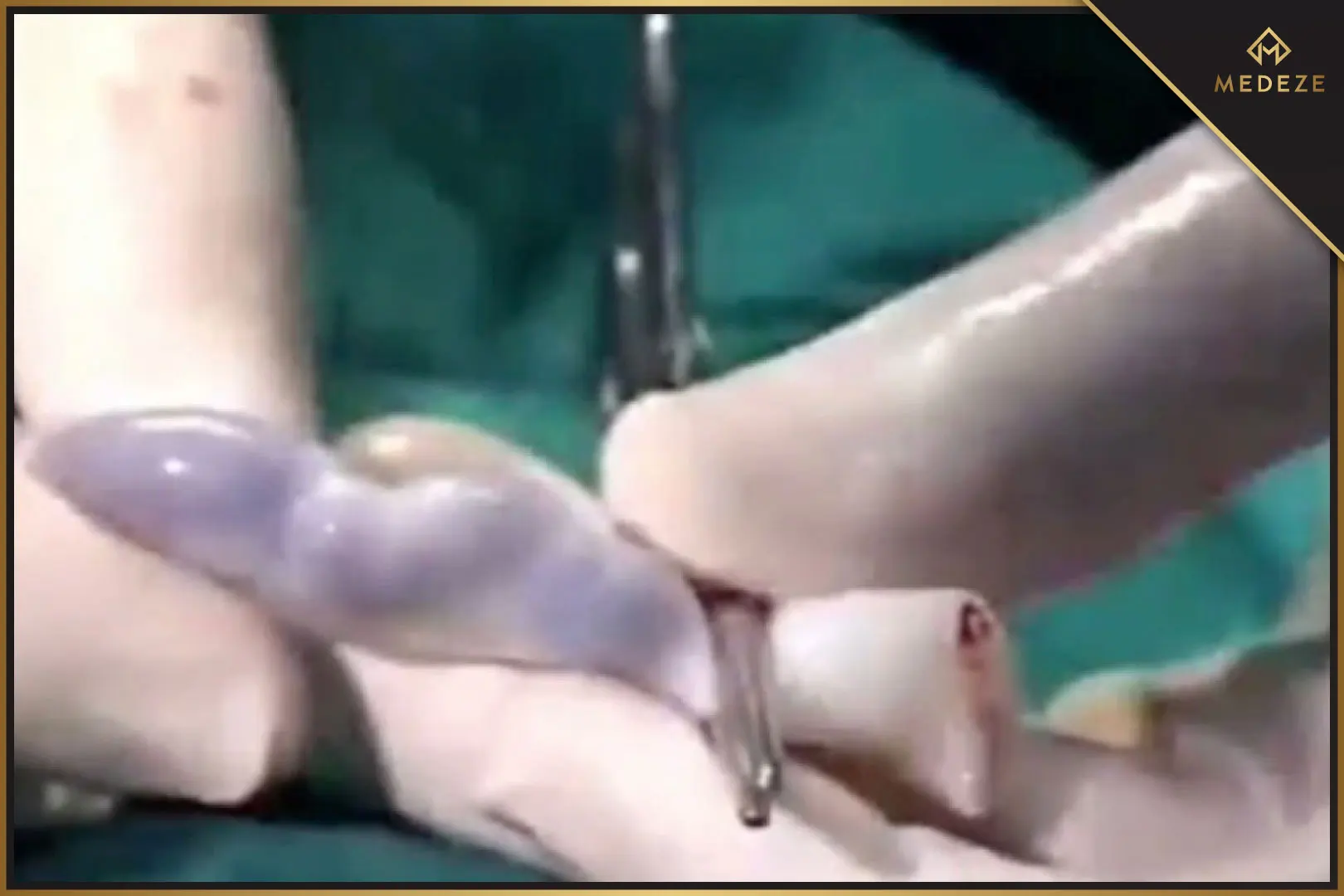
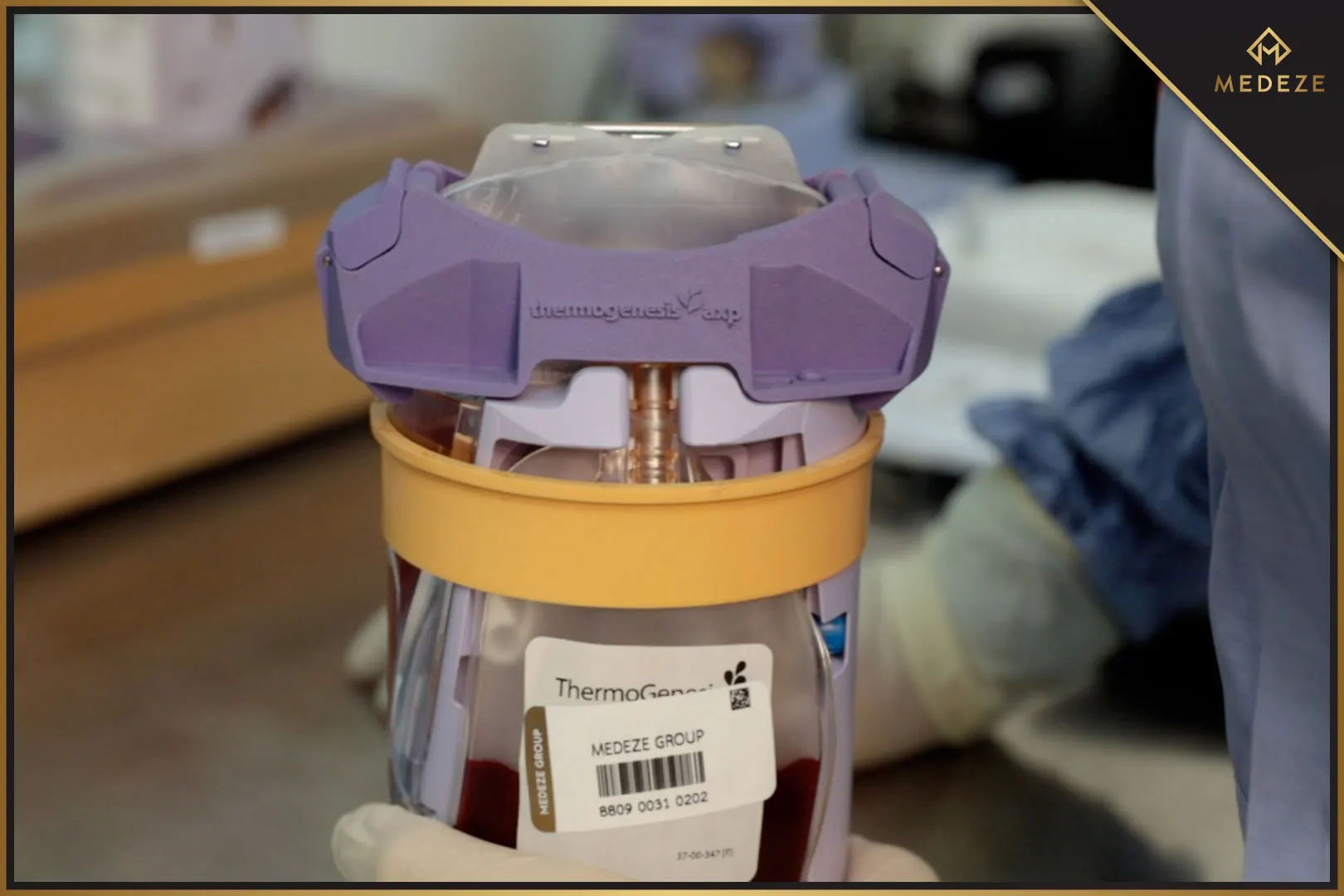
Currently, MEDEZE GROUP uses a new method called the Automatic Method for Cord Blood Extraction Process. This is considered a safe method that maintains high stem cell efficiency by collecting blood remaining in the umbilical cord and placenta at birth. Medical science has found that cord blood is full of Hematopoietic Stem Cells (HSCs), which can develop into red blood cells, white blood cells, and platelets.
For Mesenchymal Stem Cells (MSCs), since 2010, MEDEZE GROUP has developed methods for Placenta saving or Stem Cell from CORD TISSUE. This source provides a high abundance of potent stem cells and is a painless procedure. The tissue has a jelly-like characteristic called "Wharton’s jelly." This type of stem cell can develop into various cells in multiple body systems. Collection can only be done at birth, known as the Umbilical Cord Tissue Collection Process or Wharton’s Jelly Collection Procedure. Currently, only a few stem cell banks can store this specific type of tissue stem cell.
Additionally, MEDEZE GROUP can currently collect Mesenchymal Stem Cells (MSCs) from Adipose tissue (FAT). Adipose tissue is the fat located under the skin—often in areas we want to reduce—as well as around internal organs, bones, and breasts. Adipose tissue is a rich source of high-quality Mesenchymal Stem Cells (MSCs). MEDEZE GROUP utilizes techniques to collect fat samples without scarring or pain, along with technology to isolate MSCs from the fat tissue. These stem cells can be used for both medical treatment and regenerative therapy.


After the collection process, laboratory experts isolate healthy stem cells and culture them to increase their quantity over approximately one month until there are enough for use. They are then subjected to Cryopreservation at appropriate temperatures before human use. To maximize benefits, stem cell banking requires perfect isolation. Methods that freeze tissue samples without immediately isolating stem cells can damage the cells and result in low yields, potentially reducing the quality and efficacy of future treatments.
To use stem cells for injection, the cells must be cultured in a standardized laboratory, undergo rigorous testing for diseases and safety, and be properly sterilized. They must pass expert standard checks and be certified for safety by specialized physicians before they can be used for injection.
Who Can Benefit from Stem Cells?
Regarding stem cells, high-quality autologous stem cells can be stored at any age. However, for the best efficiency when used, the healthier the body is at the time of collection, the more potent the stem cells are believed to be. This is an investment in your own future health, as the risk of illness increases with age. Health is important for everyone.
Why Choose MEDEZE GROUP?

Stem cell storage with MEDEZE GROUP involves verifying both the quantity and quality of stem cells. Furthermore, MEDEZE GROUP prioritizes cleanliness at a sterile level for stem cell isolation. The entire process is controlled via a closed method for the safety and purity of the stem cells. MEDEZE GROUP collects and separates cells from the beginning of the Cryotherapy process using an automated separation system: the AXP AutoXpress Platform for Hematopoietic Stem Cells. Additionally, MEDEZE GROUP confirms the validity of the stored stem cells with a certificate displaying: Stem cell image, Total cell count, Stem cell type, Cell viability, and Number of vials, demonstrating the potential of the stored stem cells.


Moreover, MEDEZE GROUP provides a stem cell quality guarantee for up to 30 years. There is also a genetic analysis system for stem cell banking to confirm the strength and readiness of the stem cells before cryopreservation.
References
1) Hayani A, Lampeter E, Viswanatha D, Morgan D, Salvi SN. First report of autologous cord blood transplantation in the treatment of a child with leukemia. Pediatrics. 2007 Jan 1;119(1):e296-300.
2) Lee, J. S., Hong, J. M., Moon, G. J., Lee, P. H., Ahn, Y. H., & Bang, O. Y. (2010). A long‐term follow‐up study of intravenous autologous mesenchymal stem cell transplantation in patients with ischemic stroke. Stem cells, 28(6), 1099-1106.
3) Zakrzewski, W., Dobrzyński, M., Szymonowicz, M., & Rybak, Z. (2019, February 26). Stem cells: past, present, and future. Stem cell research & therapy. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6390367/.
Search Articles
Latest Articles
สายสะดือทารกแรกเกิด แหล่งรวมสเต็มเซลล์ ในการรักษาโรคและฟื้นฟูสุขภาพ
What are Mesenchymal Stem Cells (MSCs)? Exploring Their Benefits and Potential
What is Adipose Tissue? Can Fat Tissue Really Help with Anti-Aging?
Understanding Modern Innovation: NK Cells in Eliminating Cancer Cells and Virus-Infected Cells
What are NK Cells? The Immune Cells that Combat Viruses and Cancer Cell
