Get to Know a Thai Stem Cell Bank with Global-Standard Transport Capability

Get to Know a Thai Stem Cell Bank with Global-Standard Transport Capability
Medeze recognizes the potential of stem cells in treating various diseases and is determined not to disappoint those who place their trust in stem cell therapy. MEDEZE places the highest priority on service quality at every stage. This includes collection, cell isolation, preservation, and stem cell recovery for real clinical use. Each step of the process adheres strictly to international standards.
If you are looking for a laboratory that provides world-class stem cell storage services, whether for umbilical cord tissue or cord blood stem cells, More information about stem cell storage quality MEDEZE offers comprehensive solutions that exceed expectations, starting with...
Sterile collection devices and containers Sterile, temperature-controlled collection devices and containers are used to preserve tissue integrity and minimize contamination risks before delivery to the laboratory. For newborn stem cell collection, MEDEZE also provides a backup collection kit for emergencies so that parents can keep it on hand with confidence.

Tissue transport services to the laboratory Tissue transport services to the laboratory by MEDEZE staff handle the transportation of tissue directly from the hospital to the laboratory, allowing families to focus on precious moments together without concern for logistics. The MEDEZE laboratory operates 24 hours a day, ensuring that stem cell processing and storage begin immediately upon arrival.

International-standard sterile laboratory facilities International-standard sterile laboratory facilities. MEDEZE's laboratory meets Clean Room Class 100 standards for tissue culture, ensuring a sterile environment with controlled temperature and advanced air management systems to maintain optimal conditions.

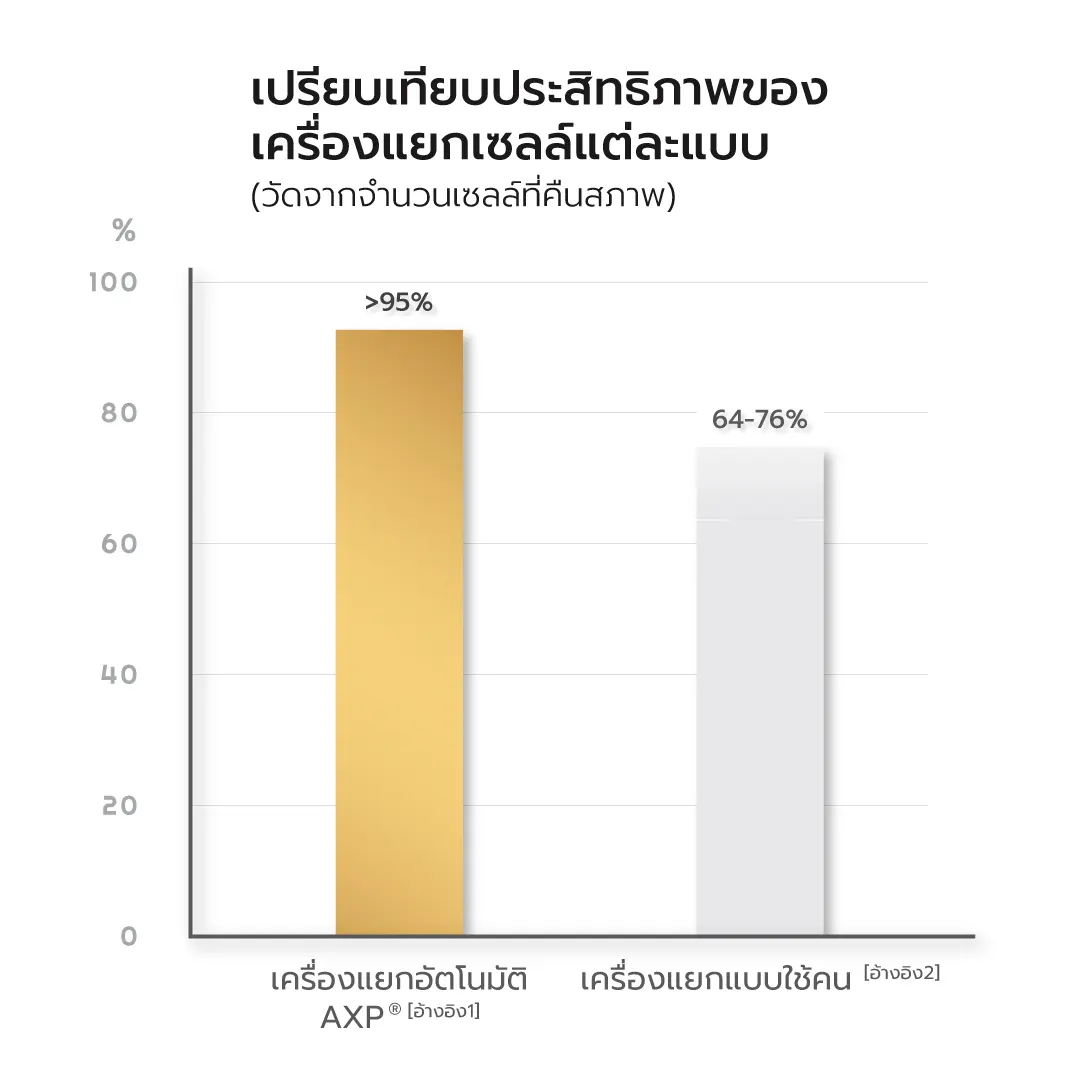
Stem cell separation process Stem cell separation at MEDEZE uses the AXP® Automated Cell Separation System, a fully automated system that reduces human error and maintains a closed system to minimize contamination. This method preserves stem cell quality far more effectively than manual separation, achieving a cell recovery rate of over 95%, compared to 64–76% with manual methods.[1,2] At the same time, the AXP® system is widely used by leading laboratories worldwide, including The New York Blood Center, the largest blood bank in the United States.[3]
Quality and quantity testing of stem cells Quality and quantity testing of stem cells after separation, stem cells undergo rigorous quality testing, including contamination screening, in accordance with AABB standards.[4] Additionally, MEDEZE includes chromosome studies as part of the quality control process for mesenchymal stem cells (MSCs).
Guaranteed cell quantity before storage Guaranteed cell quantity before storage by MEDEZE expands mesenchymal stem cells before storage to ensure a sufficient number of viable cells for future use, providing clients with added confidence.
International accreditation and certification International accreditation and certification by the American Association of Blood Banks (AABB) for newborn cord blood stem cell processing, long-term storage, and transportation for patient treatment. The laboratory also meets National Environmental Balancing Bureau (NEBB) standards for laboratory quality.

Highly experienced professionals Highly experienced professionals, MEDEZE was founded by Dr. Veerapol Khemarangsan and a team of highly specialized physiciansLearn more about MEDEZE and Dr. Veerapol and scientists with over 11 years of experience in stem cell science and tissue banking. The team is always available to guide both storage procedures and real-world clinical applications.
Quality assurance guarantee Quality assurance guarantee at MEDEZE provides a quality guarantee of up to 10 times the service value if stored stem cells cannot be used as intended, with coverage lasting up to 30 years.
Laboratory credibility and proven track record Laboratory credibility and proven track record with 11 years of experience, MEDEZE has stored stem cells for over 15,000 clients. Stem cells have already been successfully withdrawn for real-world use through the Medeze Foundation's social initiatives. Furthermore, MEDEZE has been entrusted to support seven collaborative research projects with leading medical institutions in Thailand.
References
1) Rubinstein P. Cord blood banking for clinical transplantation. Bone marrow transplantation. 2009 Nov;44(10):635-42.
2) Basford C, Forraz N, Habibollah S, Hanger K, McGuckin CP. Umbilical cord blood processing using Prepacyte‐CB increases haematopoietic progenitor cell availability over conventional Hetastarch separation. Cell proliferation. 2009 Dec;42(6):751-61.
3) Cord Blood Registry. Cord Blood Registry Achieves Industry-Leading Stem Cell Recovery With New Automation Technology. [Online]. 2007. Available at: https://www.biospace.com/article/releases/-b-cord-blood-registry-b-achieves-industry-leading-stem-cell-recovery-with-new-automation-technology-/. [July 2021]
4) AABB Accreditation Department. AABB ACCREDITED CORD BLOOD (CB) FACILITIES. [Online]. 2021. Available at: https://www.aabb.org/standards-accreditation/accreditation/accredited-facilities/cell-therapy-services/aabb-accredited-cord-blood-facilities. [July 2021]
Search Articles
Latest Articles
สายสะดือทารกแรกเกิด แหล่งรวมสเต็มเซลล์ ในการรักษาโรคและฟื้นฟูสุขภาพ
What are Mesenchymal Stem Cells (MSCs)? Exploring Their Benefits and Potential
What is Adipose Tissue? Can Fat Tissue Really Help with Anti-Aging?
Understanding Modern Innovation: NK Cells in Eliminating Cancer Cells and Virus-Infected Cells
What are NK Cells? The Immune Cells that Combat Viruses and Cancer Cell
