Stem Cells and COVID-19: Hope or Dream?

Stem Cells and COVID-19: Hope or Dream?
COVID-19 is caused by infection with the SARS-CoV-2 virus, which spreads primarily through respiratory droplets or contact with infected secretions. The respiratory epithelial cells are the main site of infection. The outbreak was first reported in Wuhan, China, in late 2019—hence the number “19” in the disease name. As cases rapidly increased worldwide, the World Health Organization (WHO) declared COVID-19 a global pandemic on March 11, 2020.[1]

Approximately 80% of infected individuals experience mild or no symptoms, similar to those of the common cold. However, a smaller proportion of patients develop severe disease (often referred to as COVID-19 pneumonia), characterized by intense inflammation and respiratory failure. Two major factors generally cause severe symptoms: damage to the respiratory system from viral infection and an excessive inflammatory response that overwhelms multiple organ systems. Current treatments mainly focus on supportive care, such as corticosteroids or anti-inflammatory medications to control inflammation, and oxygen therapy or mechanical ventilation to manage respiratory failure. Interestingly, both major complications relate to biological mechanisms that stem cells may potentially address.[2,3]
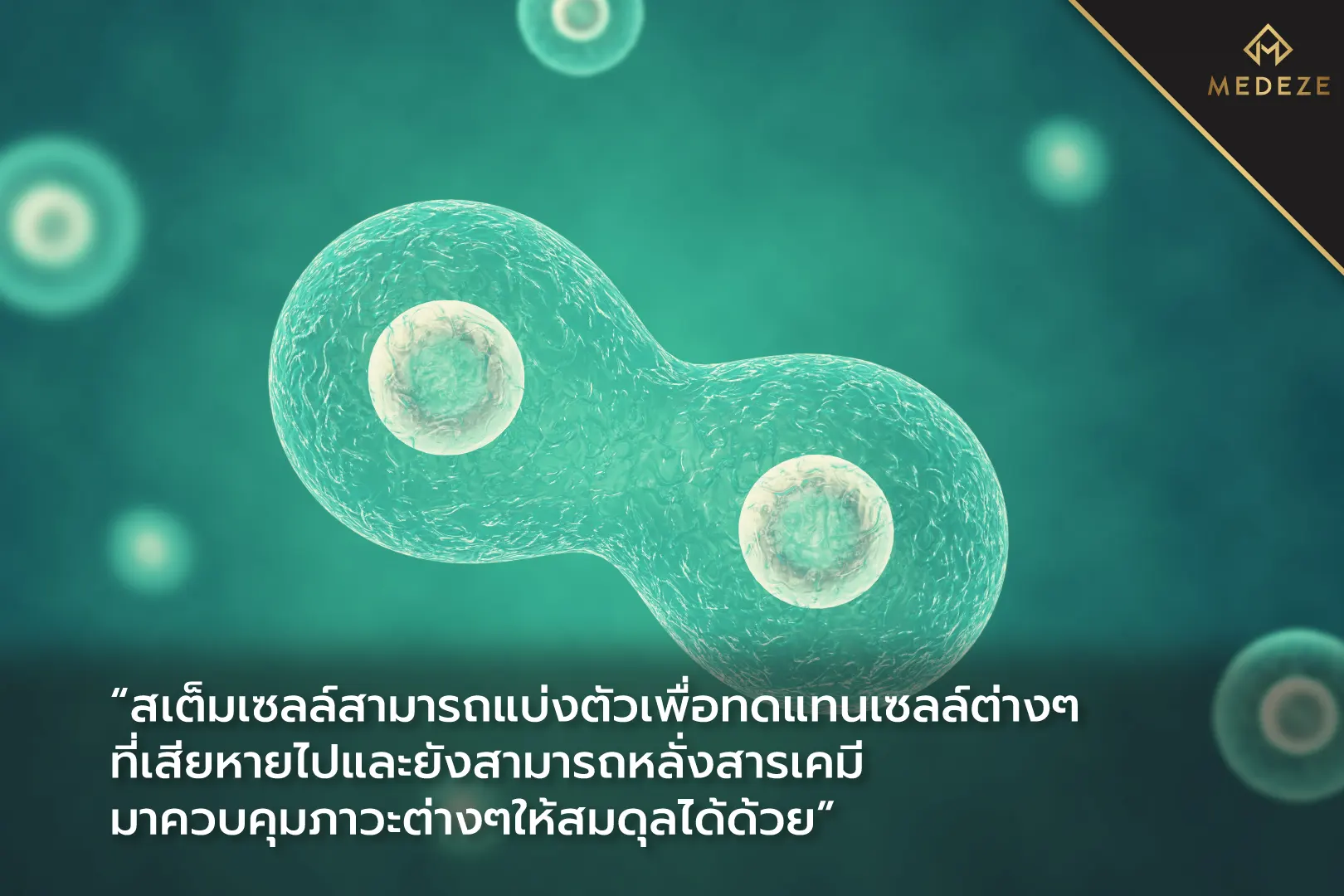
Stem cells, also known as progenitor cells, have unique characteristics that allow them to divide and differentiate into various specialized cell types. This makes them suitable for treating diseases related to cellular degeneration or tissue damage. In addition, stem cells have immunomodulatory properties, meaning they can help regulate and restore balance to the body’s biochemical environment. For this reason, stem cells are being explored for medical treatments to either stimulate or suppress abnormal biological signals that occur in certain disease conditions.
Stem cells used for medical purposes may come from donors or from a patient’s own previously stored cells. They can be collected from Bone marrow (stem cells with limited proliferation capacity) umbilical cord tissue, or adipose tissue, which provides mesenchymal stem cells with higher proliferation capacity.[5]
In the past, stem cells have been incorporated into treatments across various fields of medicine,[4] with many studies reporting encouraging clinical outcomes. During the COVID-19 pandemic, stem cells were proposed as a potential therapeutic option due to their ability to differentiate and help replace damaged respiratory cells, as well as their capacity to secrete bioactive factors that regulate excessive inflammation and restore immune balance. At present, many studies have investigated the use of various stem cell types as a therapy for COVID-19 patients, particularly in severe cases where conventional treatments have shown limited effectiveness.
For example, a study conducted in China in April 2020[6] evaluated the administration of mesenchymal stem cells at a dose of 1 million cells per kilogram of body weight in seven COVID-19 patients (including five severe cases), compared with patients who did not receive stem cell therapy. The results showed that patients who received stem cell therapy experienced clinical improvement within 2–4 days of treatment. Viral tests became negative within 7–14 days, and inflammatory markers returned to near-normal levels within six days. The researchers concluded that stem cell therapy appeared beneficial and did not result in significant adverse effects, particularly among patients with severe symptoms.

Another study conducted in the United States involved 24 patients with moderate-to-severe COVID-19 who received mesenchymal stem cell therapy. The results showed significant improvements in oxygen exchange capacity, white blood cell counts, and inflammatory markers following stem cell administration, compared with pre-treatment levels.
For those interested in stem cell banking or seeking further information, MEDEZE can be contacted directly. MEDEZE provides a team of physicians and specialists experienced in stem cell processing and cultivation to support future therapeutic applications. The company offers a range of service options and operates internationally accredited laboratory facilities, enabling stem cell collection from multiple sources with quality assurance at the time of clinical use
References
1) WHO Director-General's opening remarks at the media briefing on COVID-19 – 11 March 2020 World Health Organization (WHO) (Press release). 11 March 2020. Archived from the original on 11 March 2020. Retrieved Dec 2021.
2) Rajarshi K, Chatterjee A, Ray S. Combating COVID-19 with mesenchymal stem cell therapy. Biotechnology reports. 2020 Jun 1;26:e00467.
3) Pal D, Goyal J, Sharma U, Sharma A, Prashar S, Rathi G, Sharma B, Kumar U. Mesenchymal stem cells in SARS-CoV-2 infection: A hype or hope. Life Sciences. 2021 Nov 1;284:119901.
4) Mayo Clinic Staff. Stem cells: What they are and what they do. [Online]. June 2019. Available at: https://www.mayoclinic.org/tests-procedures/bone-marrow-transplant/in-depth/stem-cells/art-20048117. [August 2021]
5) Nair V, Talwar P, Kumar S, Chatterjee T. Umbilical cord blood transplantation and banking. Pregnancy medicine. 1st ed. Delhi: Jaypee Brothers Medical Publishers (P) Ltd. 2015:197-215.
6) Leng Z, Zhu R, Hou W, Feng Y, Yang Y, Han Q, Shan G, Meng F, Du D, Wang S, Fan J. Transplantation of ACE2-mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia. Aging and disease. 2020 Apr;11(2):216.
7) Mesenchymal stem cells derived from perinatal tissues for treatment of critically ill COVID-19-induced ARDS patients: a case series
Search Articles
Latest Articles
สายสะดือทารกแรกเกิด แหล่งรวมสเต็มเซลล์ ในการรักษาโรคและฟื้นฟูสุขภาพ
What are Mesenchymal Stem Cells (MSCs)? Exploring Their Benefits and Potential
What is Adipose Tissue? Can Fat Tissue Really Help with Anti-Aging?
Understanding Modern Innovation: NK Cells in Eliminating Cancer Cells and Virus-Infected Cells
What are NK Cells? The Immune Cells that Combat Viruses and Cancer Cell
